 If the reaction continues to sustain itself, it is said to be critical, and the mass of 235U required to produce the critical condition is said to be a critical mass. If at least one neutron from uranium-235 fission strikes another nucleus and causes it to fission, then the chain reaction will continue. Highly enriched uranium (HEU), which contains an even greater proportion of uranium-235, is sometimes used in the reactors of nuclear submarines, research reactors and nuclear weapons. Uranium enrichment removes some of the uranium-238 and increases the proportion of uranium-235. Heavy water reactors and some graphite moderated reactors can use natural uranium, but light water reactors must use low enriched uranium because of the higher neutron absorption of light water. Most neutron absorptions induce fission, though a minority result in the formation of uranium-236. For fast neutrons it is on the order of 1 barn. Its fission cross section for slow thermal neutrons is about 584.3☑ barns. It was discovered in 1935 by Arthur Jeffrey Dempster. Uranium-235 has a half-life of 703.8 million years. It is the only fissile isotope that exists in nature as a primordial nuclide. Unlike the predominant isotope uranium-238, it is fissile, i.e., it can sustain a nuclear chain reaction. Uranium-235 ( 235U or U-235) is an isotope of uranium making up about 0.72% of natural uranium. In reality, U-235 releases the 3 neutrons (it has plenty to spare anyway), and so we need to work hard to acquire the rather rare U-235 in order to harvest the power of chain reactions.Uranium metal highly enriched in uranium-235 It is just an unfortunate coincidence that U-238 has 3 more neutrons than U-235, and the textbook uranium chain reaction involves releasing 3 free neutrons, so people think U-238 is the fissile material. And it is easier to slow down too-fast neutrons (this is what moderators do) than to speed up too-slow ones. Moreover, it often releases multiple neutrons when breaking up. U-235, on the other hand, is very sensitive to being broken up by slow neutrons (almost 600 times more sensitive according to Wikipedia). 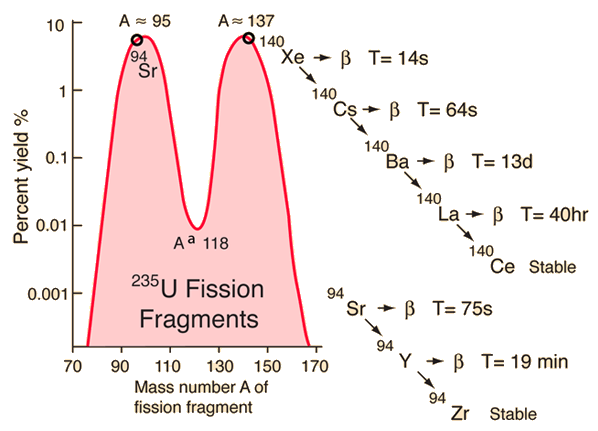
You can break it with a fast neutron, but it won't release enough fast neutrons for this to continue. 
The requirement for a material to be fissile (to be able to sustain a chain fission reaction) is not simply that it have neutrons, but that each fission releases enough neutrons of the right energy to trigger further reactions.Īs it so happens (and I know of no simple reason for this, it's just the way the equations work), U-238 isn't fissile. All neutrons can cause fission in U-235, so its reaction is sustainable.Since too few neutrons are born from fission at the energy required to fission U-238 (and other non-fissile isotopes), a reaction with only U-238 is not sustainable.It also shows that any neutron energy can cause fission in U-235.The plot in the link shows that only very high energy neutrons can cause fission in U-238 (far right brown line)(note the logarithmic axes), which correlates the critical energy concept only neutrons above a certain energy are bringing enough to the U-238 nucleus to get it above its critical energy.Low critical energy and being fissile is generally linked to odd neutron counts.In fissionable and non-fissile isotopes, like U-238, critical energy is greater than the excitation of the nucleus when it has absorbed a neutron, so the neutron must bring additional energy to cause fission.So any absorbed neutron can cause fission in these nuclei, though they still have a capture probability (U-235 can absorb a neutron and become U-236 instead of fissioning). 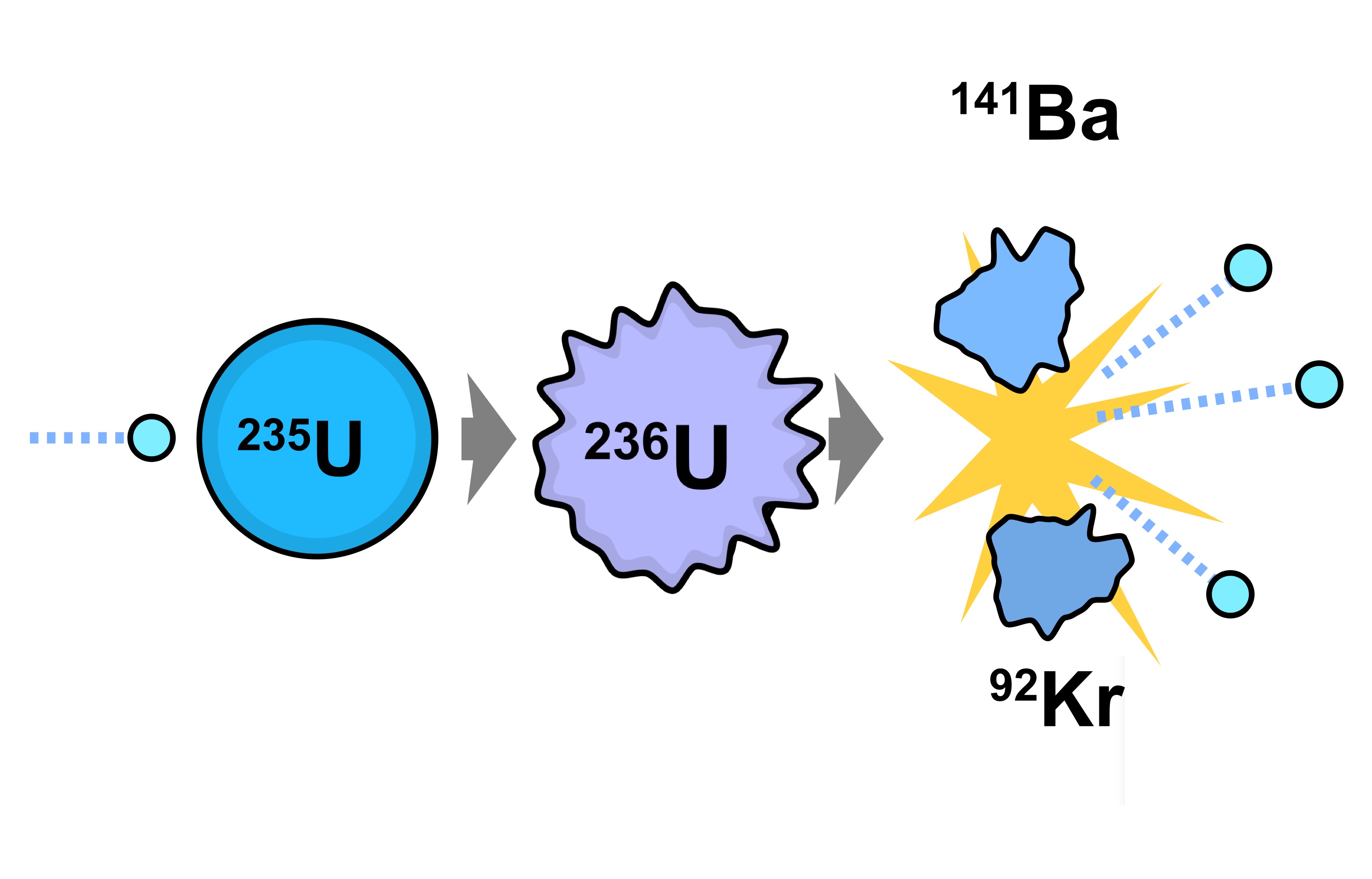
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
